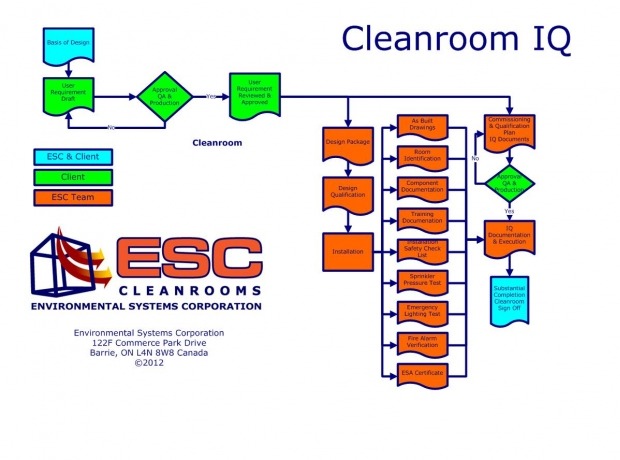
The Installation Qualification is the proving we have installed what we initially intended to, or have a detailed log of the changes and rationale for the change.
The Operational Qualification is proving that the design can consistently function in day to day use.
The Performance Qualification is how we handle the changes in operational status, which can happen with any system, and prove acceptance to start production.
This ends the construction validation process and turns over the system to the operations group.
At ESC we can assist you in understanding this process and can provide as much assistance as is required to ensure completion of the process.